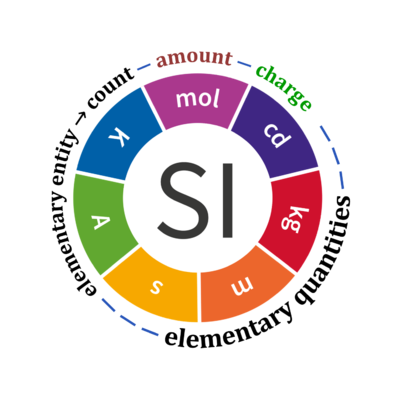
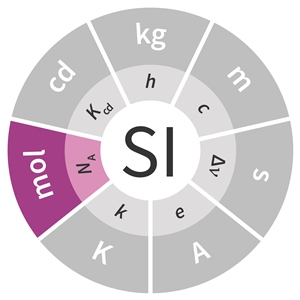
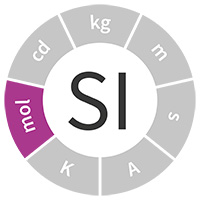
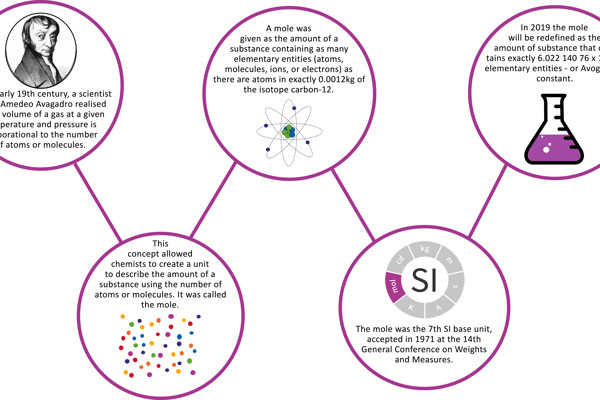
Chem Notes Mole- SI unit for amount of matter Mole- SI unit for amount of matter mol 6.02 X representative particles= Avogadro's Number. - ppt download

Chapter 7 Chemical Quantities. The Mole SI base unit to measure the amount of a substance 1 mole of anything = x representative particles. - ppt download
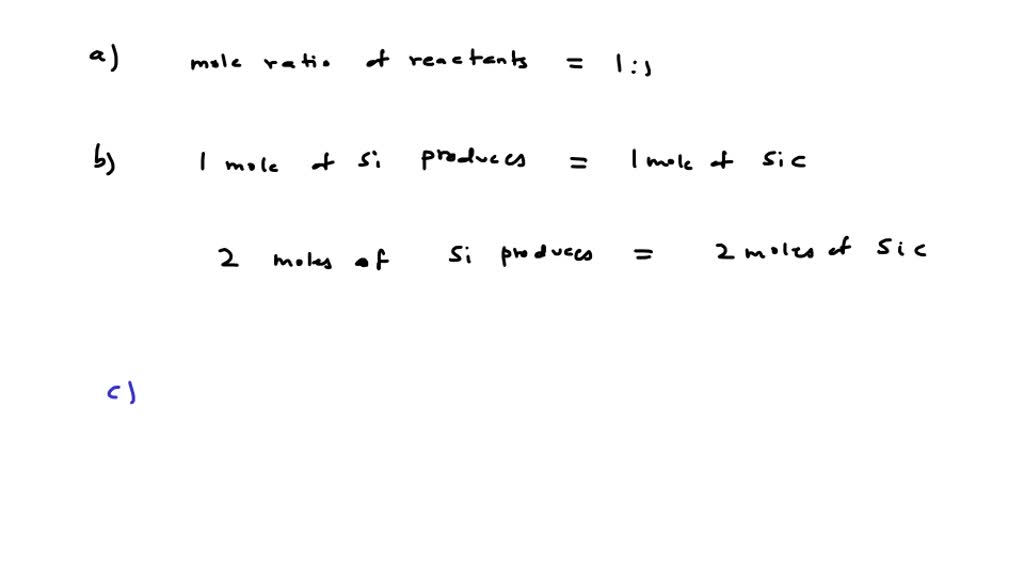
SOLVED: 'Consider the reaction: Si + € 3 Sic What is the mole ratio of the reactants? If 2 moles of Si were consumed in the reaction; how many moles of SiC